Law & Regulation
Algernon sells Ifenprodil, zooms in on DMT for stroke care
The upcoming study will compare the effects of DMT and a placebo in stroke patients.
The post Algernon sells Ifenprodil, zooms in on DMT for stroke care…
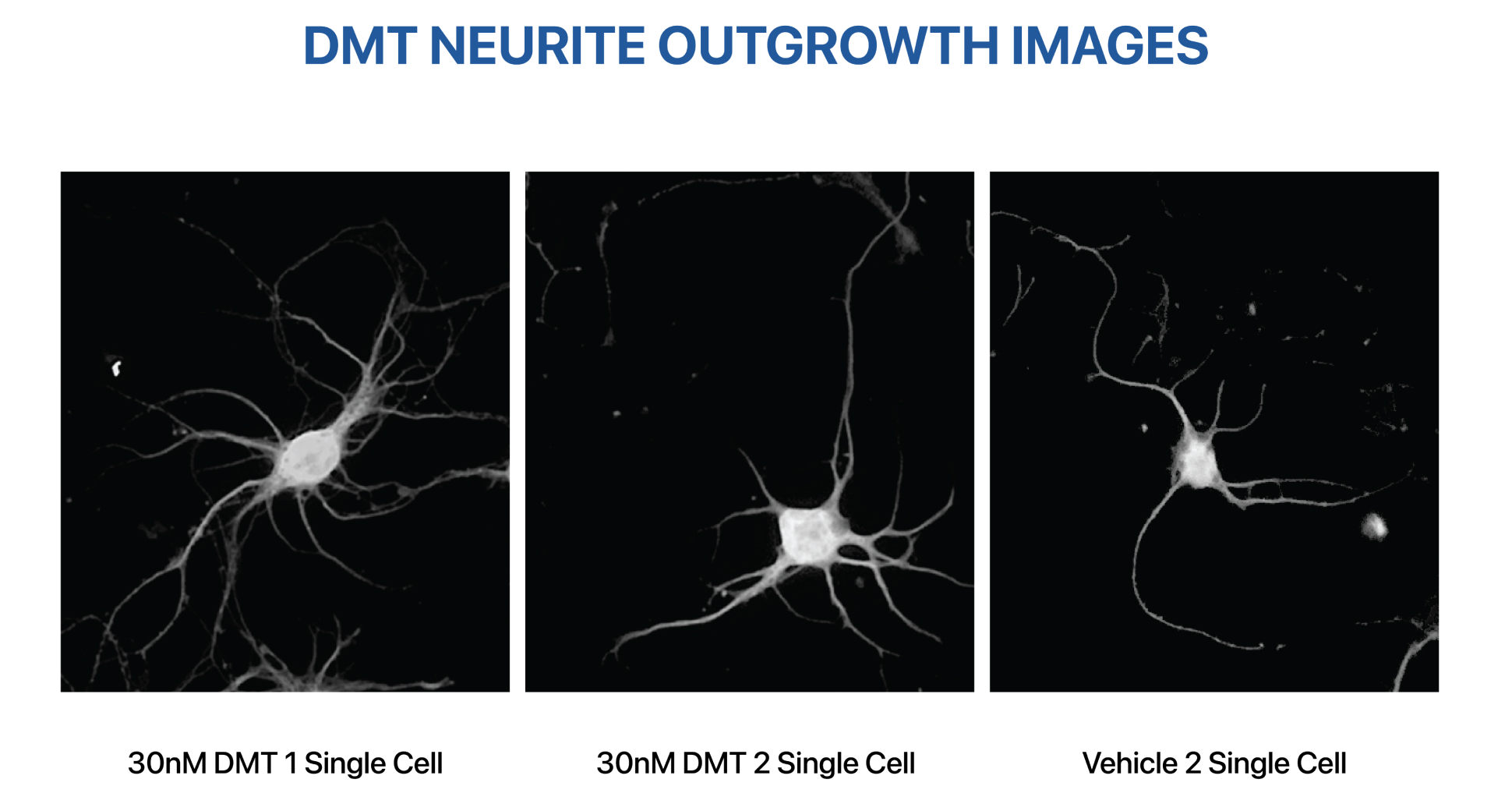
Algernon Pharmaceuticals Inc. (CSE: AGN) (OTCQB: AGNPF) announced Monday that it will prioritize the development of its intravenous formulation of AP-188 (DMT) to see if it can help patients recover from strokes.
The decision follows the Canadian clinical-stage pharmaceutical’s Wednesday sale of its Ifenprodil research program to Seyltx, a U.S.-based private drug developer, for $2 million in cash and a 20% equity stake in Seyltx.
Algernon’s subsidiary, Algernon NeuroScience, will lead the research and development of the DMT stroke program. The subsidiary already completed a feasibility study and finalized the design for a 40-patient Phase 2a clinical trial, which will investigate the use of a sub-psychedelic dose of DMT administered intravenously to patients hospitalized after suffering an acute ischemic stroke.
“Investigating neuroplasticity in a clinical setting, as a potential new treatment approach for ischemic stroke patients, is a new and promising area of research,” Algernon CEO Christopher J. Moreau said in a statement. “Algernon is a global leader in this important area of research, and we look forward to further advancing the stroke program through our planned Phase 2a DMT Stroke study.”
The Phase 2a study will primarily focus on safety while also gathering data on efficacy measures such as brain tissue preservation, motor recovery, depression, and various biomarkers associated with stroke pathophysiology.
The choice to move forward with the research was encouraged by earlier small-scale studies that showed DMT could induce brain repair effects at non-psychedelic doses during a six-hour treatment session.
“The amount given exceeded the human equivalent of the dose used in preclinical studies in rats, which demonstrated neuroprotective effects,” the company said.
The post Algernon sells Ifenprodil, zooms in on DMT for stroke care appeared first on Green Market Report.
research psychedelic dmt
-

 Psilocybin6 days ago
Psilocybin6 days agoPassover Perspectives: Psychedelics, Moses, and the Burning Bush
-

 Psychedelics5 days ago
Psychedelics5 days agoAlgernon NeuroScience and the Centre for Human Drug Research to Present DMT Phase 1 Stroke Clinical Data at the Interdisciplinary Conference on Psychedelic Research June 6 – 8th, 2024
-

 Psychedelics6 days ago
Psychedelics6 days agoRevive Therapeutics Announces Type C Meeting Request Granted by FDA for Clinical Study of Bucillamine to Treat Long COVID
-

 Psychedelics5 days ago
Psychedelics5 days agoatai Life Sciences Announces Dosing of First Patient in Part 2 of Beckley Psytech’s Phase 2a Study Exploring BPL-003 Adjunctive to SSRIs in Patients with Treatment Resistant Depression
-

 Law & Regulation6 days ago
Law & Regulation6 days agoTryp Therapeutics merger with Exopharm approved by shareholders
-

 Psychedelics6 days ago
Psychedelics6 days agoSilo Pharma Announces Positive Results for Intranasal PTSD Treatment
-

 Psychedelics6 days ago
Psychedelics6 days agoOptimi Health and Kwantlen Polytechnic University Applied Genomics Centre Partner to Advance Mushroom Science and Research
-

 Psilocybin6 days ago
Psilocybin6 days agoPsilocybin and Meditation: Unlocking the Potential for Enhancing Insightfulness










