Public Companies
Healthcare Co.’s To Develop Fast-Dissolving MDMA Tablet
Source: Streetwise Reports 09/12/2022
One biopharma co. is teaming with a much larger pharmaceutical company to develop a fast-dissolving…
Source: Streetwise Reports 09/12/2022
One biopharma co. is teaming with a much larger pharmaceutical company to develop a fast-dissolving oral tablet to deliver MDMA to patients with Alcohol Use Disorder. Find out why developing an oral tablet could benefit the company in the long run.Adding to a string of recent announcements designed to increase and protect its intellectual property (IP), Awakn Life Sciences Corp. (AWKN:NEO; AWKNF:OTCQB) has signed a drug development agreement with Catalent Inc. (CTLT:NYSE), a provider of advanced delivery technologies for drugs, among other things.
The duo plan to develop a fast-dissolving oral tablet that will make it easier for Awakn patients to take the company's MDMA formula during clinic visits and clinical trials. It also provides a defined pathway toward regulatory approval, which is the goal of the program.
There is already an oral MDMA tablet that enters the bloodstream by dissolving in the stomach, but the tablet both companies are developing would enter the bloodstream by dissolving on the tongue.
Awakn chose to work with Catalent primarily due to its proprietary Zydis orally dissolving tablet.
Zydis is a freeze-dried solid that instantly disperses in the mouth — without any additional water.
Awakn plans to use Zydis in its late-stage MDMA-assisted therapy clinical trials, as it has the potential to get the drug into the patient significantly faster and therefore shorten the treatment time, making it more attractive to clinicians.
Tom Hawkeswood, president and division head of pharma product delivery with Catalent, said Zydis technology provides "multiple potential benefits including initial acceptance, adherence, convenience, and advantages for products where a rapid onset of action is possible."
He added, "Catalent has a proven track record of working with partners to develop, launch and supply new therapies that are differentiated from traditional and generic dose forms and that are difficult to emulate."
Awakn has the exclusive rights to use Zydis technology for addiction — that means no other drug company can use Zydis to treat addictive behaviors. That includes substance and behavioral addictions, as well as other mental health disorders, including anxiety, depression, Post Traumatic Stress Disorder, as well as eating disorders.
Awakn's Phase 2a clinical trial, which examined MDMA-assisted therapy as a treatment for Alcohol Use Disorder, was positive.
The trial results were published in The Journal of Psychopharmacology in February 2021 and demonstrated the potential for MDMA-assisted therapy to be used as an effective treatment for AUD.
Awakn's MDMA treatment for AUD is in addition to its lead program — Ketamine-assisted psychological therapy for AUD.
Partheniou has a Speculative Buy rating on Awakn with a 12-month target of CA$8 per share.
It is estimated that over 400 million people globally suffer from AUD, and only 16% of those suffering seek out treatment. Unfortunately, even for those who do pursue treatment, relapse rates remain consistently high, with 75% resuming dangerous levels of alcohol consumption within 12 months of treatment.
A new, more effective treatment for AUD is desperately needed, particularly given the increase in alcohol dependence following the COVID-19 global pandemic.
"This agreement further strengthens the IP moat for our MDMA program. Having the right partner to help facilitate our upcoming Phase IIb clinical trial is key," Awakn CEO Anthony Tennyson said.
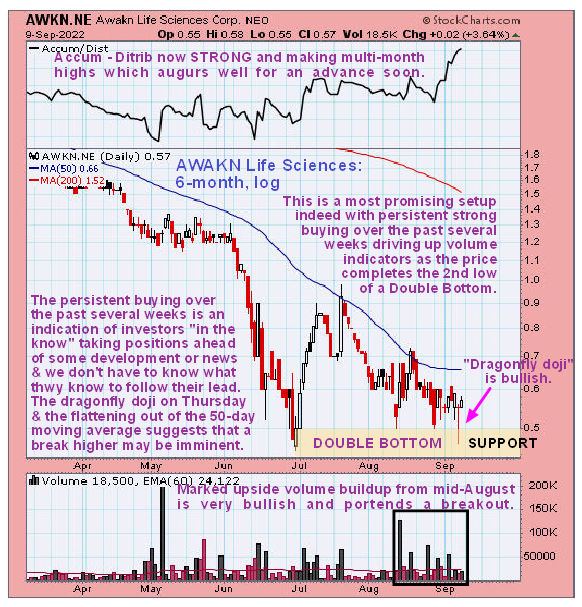
In a September 11, 2022 post on clivemaund.com, Technical Analyst Clive Maund was bullish on the news.
"There is a reason for banging on about AWKN Life Sciences now, that reason being that its technical setup is exceptionally positive and keeps getting better. On its latest 6-month chart, we can see that it is in the process of completing the second low of a Double Bottom and also that the volume pattern is strongly positive with persistent heavy buying since mid-August," Maund wrote in a recent post. "This is a sign of determined accumulation by buyers who "know something" and we don't have to know what they know; simply follow their footprints."
He added, "AWAKN is therefore considered to be an even stronger Buy than when we last looked at it, and this is one that it is considered to be worth going overweight on."
Recent Deals
In August, Awakn signed an out-licensing partnership deal with Knoxville, Tennessee-based Revitalist Lifestyle and Wellness Ltd. (RVLWF:OTCMKTS;CALM:CSE; ) to bring its ketamine-assisted therapy to Revitalist's 10 U.S.-based ketamine wellness clinics.
The company followed the Revitalist deal with another agreement with Burlington, Ontario-based Wellbeings Pain Management and Dependency Clinic, which will be the first company in Canada to deliver Awakn's off-label treatment. Both deals will see Awakn receive annual fees, revenue-sharing on a per-treatment basis, and patient and treatment data.
On August 16, 2022, Stifel GMP Analyst Andrew Partheniou published a note highlighting Awakn's new licensing business.
"These deals and others to come have the potential to generate "attractive capex-light, high-margin software-as-a-service-like recurring revenues, which investors are currently getting for free at Awakn's current valuation," Partheniou wrote.
Partheniou has a Speculative Buy rating on Awakn with a 12-month target of CA$8 per share.
Catalysts
For its lead program — ketamine-assisted psychological therapy for AUD — Awakn is working on its regulatory authority and ethics committee review in the U.K., followed by the clinical trial phase, all with the aim of receiving marketing authorization in the U.K. by 2026.
Once that's achieved, ketamine-assisted therapy could be prescribed by a doctor and covered by healthcare insurance plans. It could also lead to other revenue streams like royalty payments and higher licensing fees.
Regulatory approval by the Food and Drug Administration will likely come sometime in 2027, but Awaken has hired U.S. consultant Veristat to map out the most efficient route to U.S. regulatory approval for its lead program.
"AWAKN is therefore considered to be an even stronger Buy than when we last looked at it, and this is one that it is considered to be worth going overweight on."
— Technical Analyst Clive Maund
Awakn recently applied for the Innovative Licensing and Access Pathway (ILAP) program in the U.K. for its ketamine-assisted psychotherapy. The program helps accelerate the time to market medicines and treatments. The company should get a response in October.
In May, the biotech applied for a patent cooperation treaty or PCT — a global patent application — for its proprietary ketamine-assisted treatment, which would give it some protection from companies attempting to copy it. CEO Anthony Tennyson hopes to get a positive opinion on the PCT application before the end of the year.
Share Structure and Institutions
Awakn has about CA$2 million cash in the treasury and will soon need to raise more.
Awakn's biggest shareholder is OrbiMed Advisors LLC. OrbiMed owns 8.93% or about 2.4 million shares.
Other institutional holders include: London-based Psych Capital; New York-based JLS Fund; investment fund Palo Santo; Florida-based Iter Investments; Grand Cayman, Cayman Islands-based Negev Capital; San Juan, Puerto Rico-based Ambria Capital; and London-based Neo Kuma Ventures.
AWKN has issued almost 27 million shares, and all but roughly 6 million shares remain tightly held. The company trades in a 52-week range of CA$0.45 and CA$3.36.
Awakn Life Sciences researches, develops, and commercializes therapeutics to treat substance and behavioral addictions. Awakn's near-term focus is on AUD, more commonly known as alcoholism.
[SMNLINSERT]
Disclosures:
1) Brian Sylvester wrote this article for Streetwise Reports LLC and provides services to Streetwise Reports as an independent contractor. He or members of his household own securities of the following companies mentioned in the article: None. He and members of his household are paid by the following companies mentioned in this article: None. His company has a financial relationship with the following companies referred to in this article: None.
2) The following companies mentioned in this article are billboard sponsors of Streetwise Reports: Awakn Life Sciences Corp. Click here for important disclosures about sponsor fees. The information provided above is for informational purposes only and is not a recommendation to buy or sell any security.
3) The article does not constitute investment advice. Each reader is encouraged to consult with his or her individual financial professional and any action a reader takes as a result of information presented here is his or her own responsibility. By opening this page, each reader accepts and agrees to Streetwise Reports' terms of use and full legal disclaimer. This article is not a solicitation for investment. Streetwise Reports does not render general or specific investment advice and the information on Streetwise Reports should not be considered a recommendation to buy or sell any security. Streetwise Reports does not endorse or recommend the business, products, services or securities of any company mentioned on Streetwise Reports.
4) From time to time, Streetwise Reports LLC and its directors, officers, employees or members of their families, as well as persons interviewed for articles and interviews on the site, may have a long or short position in securities mentioned. Directors, officers, employees or members of their immediate families are prohibited from making purchases and/or sales of those securities in the open market or otherwise from the time of the decision to publish an article until three business days after the publication of the article. The foregoing prohibition does not apply to articles that in substance only restate previously published company releases. As of the date of this article, officers and/or employees of Streetwise Reports LLC (including members of their household) own securities of Awakn Life Sciences Corp., a company mentioned in this article.
5) This article does not constitute medical advice. Officers, employees and contributors to Streetwise Reports are not licensed medical professionals. Readers should always contact their healthcare professionals for medical advice
( Companies Mentioned: AWKN:NEO; AWKNF:OTCQB, CTLT:NYSE, RVLWF:OTCMKTS;CALM:CSE; , )
mdma ketamine therapeutics therapy psychotherapy depression anxiety alcoholism investors shares fund cse negev capital deal capital awakn life sciences awakn revitalist lifestyle and wellness revitalist authorization
-

 Psilocybin1 week ago
Psilocybin1 week agoCalifornia advances bill for psychedelics centers
-

 Psilocybin6 days ago
Psilocybin6 days agoPassover Perspectives: Psychedelics, Moses, and the Burning Bush
-

 Psychedelics1 week ago
Psychedelics1 week agoPsychedelics Can Offer More Than Therapy On Its Own
-

 Psychedelics5 days ago
Psychedelics5 days agoAlgernon NeuroScience and the Centre for Human Drug Research to Present DMT Phase 1 Stroke Clinical Data at the Interdisciplinary Conference on Psychedelic Research June 6 – 8th, 2024
-

 Psychedelics6 days ago
Psychedelics6 days agoRevive Therapeutics Announces Type C Meeting Request Granted by FDA for Clinical Study of Bucillamine to Treat Long COVID
-

 Psychedelics5 days ago
Psychedelics5 days agoatai Life Sciences Announces Dosing of First Patient in Part 2 of Beckley Psytech’s Phase 2a Study Exploring BPL-003 Adjunctive to SSRIs in Patients with Treatment Resistant Depression
-

 Law & Regulation5 days ago
Law & Regulation5 days agoTryp Therapeutics merger with Exopharm approved by shareholders
-

 Psychedelics6 days ago
Psychedelics6 days agoSilo Pharma Announces Positive Results for Intranasal PTSD Treatment






