Psilocybin
Psilocybin-like drug may help people suffering from anorexia: COMPASS study
The majority of participants reported significant improvements in their overall mental health
The post Psilocybin-like drug may help people suffering…
Around 40 per cent of people who took a psilocybin-like drug developed by COMPASS Pathways plc (Nasdaq: CMPS) reported reduced symptoms of anorexia after a single dose but 90 per cent felt one dose was not enough.
That is according to a study from the United Kingdom-based biotechnology published in the prominent journal Nature Medicine on Monday. The COMP360 drug is an investigational synthetic formulation of psilocybin developed by the company.
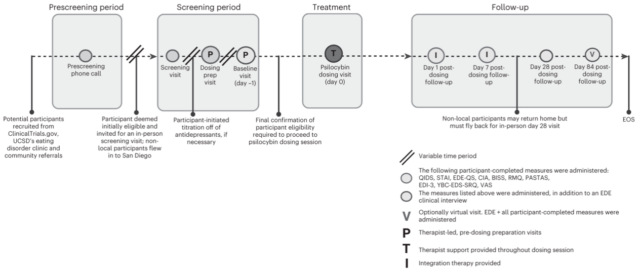
The research was conducted among 10 adult female participants who received a single 25-milligram dose of the psilocybin drug. The company believes this is the first clinical research study to report the impact of psilocybin treatment on people suffering from the disorder.
It found that 40 per cent of participants had clinically significant reductions in eating disorder psychopathology three months later. It also found that 8 out of 10 felt the experience was one of the most meaningful in their lives.
Eating disorder psychopathology refers to the psychological and behavioural aspects of eating disorders, such as severely limiting food intake for fear of becoming overweight among those who suffer from anorexia.
Additionally, 70 per cent said taking COMP360 caused a significant shift in their personal identity and quality of life.
Read more: PharmAla Biotech receives guidance on new MDMA-like drug for PTSD and Autism
Read more: Filament Health to list on Nasdaq through new business arrangement
Despite some positive results, 9 out of 10 participants said that one dose was not enough. A total of 158 women expressed an interest in participating in the study but only 10 ultimately met the necessary requisites from the Diagnostic and Statistical Manual of Mental Disorders.
The manual is a publication by the American Psychiatric Association detailing the criteria necessary for diagnosing mental health disorders. It contains written contributions from over 200 experts in the field of mental health and information on over 70 disorders. Each disorder described in the document has a set of symptoms that must be present for its diagnosis along with conditions that must be ruled out.
The psilocybin therapy facilitated by trained therapists was found to be safe and well-tolerated overall among all participants. Five out of 10 patients were found to have gained weight during the study’s three-month follow up but COMPASS says the gains were not statistically significant.
The study on the drug was held at the University of California San Diego School of Medicine last year. It was conducted by professors Walter H. Kaye and Stephanie Knatz Peck from the school’s Eating Disorder Center.
The data was originally presented at the Society of Biological Psychiatry’s annual meeting in Louisiana last year prior to being published in Nature.
“This study shows promising preliminary evidence that COMP360 psilocybin treatment could help people living with this difficult-to-treat condition. We are now looking to investigate these findings further in our larger phase 2 study,” said Guy Goodwin, Chief Medical Officer at COMPASS.
“Our findings suggest that a subset of people experienced significant improvements in eating disorder psychopathology. Our hope is that this study provides a pathway to continue to find better ways to address the psychology of anorexia from the inside out,” said Knatz Peck.
Study design and timeline. Image via Nature Medicine
Read more: PharmaTher Holdings finishes testing microneedle DMT patch
Read more: COMPASS Pathways reports 52% YoY cash loss
COMP360 is used for treating depression as well
The biotech company recently secured a US$50 million loan from Hercules Capital, Inc. (NYSE: HTGC) to help support its Phase III COMP360 program for treatment-resistant depression (TRD).
At the end of May, COMPASS released data showing that the psilocybin drug was also efficacious for the treatment of depression in cancer patients — with over 50 per cent of participants who took one dose of COMP360 experiencing a remission in depression symptoms for 18 months.
The drug has been designated as a breakthrough therapy by the United States Food and Drug Administration (FDA), an organization that recently published its first draft guidance document on psychedelics research.
COMPASS reported having a cash position of US$117.1 million at the end of Q2 this year, a 52 per cent drop year-over-year from the second quarter of 2022. The London U.K.-based company also has U.S. offices in San Francisco and New York.
The company’s shares rose by 4.1 per cent Friday to US$9.13 on the Nasdaq exchange and have risen by over 18 per cent since the beginning of this year.
rowan@mugglehead.com
The post Psilocybin-like drug may help people suffering from anorexia: COMPASS study appeared first on Mugglehead Magazine.
psilocybin mdma dmt psychiatry therapy breakthrough therapy depression ptsd psychedelics shares nasdaq capital pharmather compass filament health filament fda research
-

 Psilocybin6 days ago
Psilocybin6 days agoPassover Perspectives: Psychedelics, Moses, and the Burning Bush
-

 Psychedelics5 days ago
Psychedelics5 days agoAlgernon NeuroScience and the Centre for Human Drug Research to Present DMT Phase 1 Stroke Clinical Data at the Interdisciplinary Conference on Psychedelic Research June 6 – 8th, 2024
-

 Psychedelics6 days ago
Psychedelics6 days agoRevive Therapeutics Announces Type C Meeting Request Granted by FDA for Clinical Study of Bucillamine to Treat Long COVID
-

 Psychedelics5 days ago
Psychedelics5 days agoatai Life Sciences Announces Dosing of First Patient in Part 2 of Beckley Psytech’s Phase 2a Study Exploring BPL-003 Adjunctive to SSRIs in Patients with Treatment Resistant Depression
-

 Law & Regulation6 days ago
Law & Regulation6 days agoTryp Therapeutics merger with Exopharm approved by shareholders
-

 Psychedelics6 days ago
Psychedelics6 days agoSilo Pharma Announces Positive Results for Intranasal PTSD Treatment
-

 Psychedelics6 days ago
Psychedelics6 days agoOptimi Health and Kwantlen Polytechnic University Applied Genomics Centre Partner to Advance Mushroom Science and Research
-

 Psychedelics6 days ago
Psychedelics6 days agoRevive Therapeutics Announces Type C Meeting Request Granted by FDA for Clinical Study of Bucillamine to Treat Long COVID










